
2023.09.18
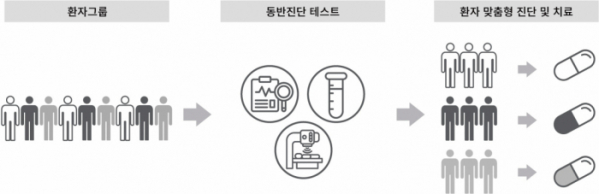 Dx&Vx (DXVX) announced on September 18 that it will enter the companion diagnostics market by launching an integrated service utilizing various types of gene panels.
Companion diagnostics is a diagnostic technique that predicts whether a particular drug will work for a patient. In 2014, the US Food and Drug Administration (FDA) recommended the use of companion diagnostics in clinical trials and issued mandatory guidelines for new drug development and companion diagnostics.
It has been reported that incorporating companion diagnostics into new drug development can triple its success rate and reduce the development costs to one-third. Accordingly, a number of pharmaceutical companies in Korea and abroad have begun jointly developing companion diagnostics for target biomarkers with diagnostic companies from the early stages of drug development to the clinical and commercialization stages.
Dx&Vx plans to target the market with differentiated companion diagnostic products and services encompassing a wide variety of existing companion diagnostic techniques such as next-generation sequencing (NGS), fluorescence in situ hybridization (FISH), and polymerase chain reaction (PCR) by leveraging its know-how in genome analysis. With an AI-based digital database system with more than 400,000 pieces of clinical genomic analysis data as well as expertise in developing and manufacturing validated PCR and immunodiagnostic kits, the company is capable of providing customized services that cater to the needs of client companies with respect to the cell tissues and blood samples required for companion diagnostics.
Also, with global clinical research and RA experts who recently joined the company, Dx&Vx can provide integrated, all-in-one companion diagnostic development solutions that are differentiated from those of other companies.
A company official said, “We’ll lead the market with revolutionary and integrated companion diagnostic services based on the genomic analysis know-how and clinical analysis data on genes related to rare diseases, cancer, and various adult diseases that we’ve accumulated for years. […] We will expand our companion diagnostic service clients from hospitals and clinics nationwide to include research institutes and pharmaceutical companies and expand our services to the global market in the future.”
Dx&Vx is currently receiving development requests from startups, biotech ventures, and biopharmaceutical companies developing anti-cancer drugs and is poised to launch services for overseas biopharmaceutical companies as well. It is also in talks with Professor Antonio Gasbarrini from Gemelli Hospital in Italy to operate a local companion diagnostic center.
According to a report by global research firm, Technavio, the companion diagnostics market is project to grow to USD 8.3 billion (approx. KRW 10 trillion) in size by 2024, with a compound annual growth rate of more than 26%.
Source: etoday
Dx&Vx (DXVX) announced on September 18 that it will enter the companion diagnostics market by launching an integrated service utilizing various types of gene panels.
Companion diagnostics is a diagnostic technique that predicts whether a particular drug will work for a patient. In 2014, the US Food and Drug Administration (FDA) recommended the use of companion diagnostics in clinical trials and issued mandatory guidelines for new drug development and companion diagnostics.
It has been reported that incorporating companion diagnostics into new drug development can triple its success rate and reduce the development costs to one-third. Accordingly, a number of pharmaceutical companies in Korea and abroad have begun jointly developing companion diagnostics for target biomarkers with diagnostic companies from the early stages of drug development to the clinical and commercialization stages.
Dx&Vx plans to target the market with differentiated companion diagnostic products and services encompassing a wide variety of existing companion diagnostic techniques such as next-generation sequencing (NGS), fluorescence in situ hybridization (FISH), and polymerase chain reaction (PCR) by leveraging its know-how in genome analysis. With an AI-based digital database system with more than 400,000 pieces of clinical genomic analysis data as well as expertise in developing and manufacturing validated PCR and immunodiagnostic kits, the company is capable of providing customized services that cater to the needs of client companies with respect to the cell tissues and blood samples required for companion diagnostics.
Also, with global clinical research and RA experts who recently joined the company, Dx&Vx can provide integrated, all-in-one companion diagnostic development solutions that are differentiated from those of other companies.
A company official said, “We’ll lead the market with revolutionary and integrated companion diagnostic services based on the genomic analysis know-how and clinical analysis data on genes related to rare diseases, cancer, and various adult diseases that we’ve accumulated for years. […] We will expand our companion diagnostic service clients from hospitals and clinics nationwide to include research institutes and pharmaceutical companies and expand our services to the global market in the future.”
Dx&Vx is currently receiving development requests from startups, biotech ventures, and biopharmaceutical companies developing anti-cancer drugs and is poised to launch services for overseas biopharmaceutical companies as well. It is also in talks with Professor Antonio Gasbarrini from Gemelli Hospital in Italy to operate a local companion diagnostic center.
According to a report by global research firm, Technavio, the companion diagnostics market is project to grow to USD 8.3 billion (approx. KRW 10 trillion) in size by 2024, with a compound annual growth rate of more than 26%.
Source: etoday